The landscape of recombinant protein manufacturing is evolving rapidly, with an increased emphasis on high-concentration formulations to meet clinical demands. Companies venturing into this arena face the challenge of optimizing protein process development while ensuring compliance with regulatory standards and maintaining product quality.
Comprehensive Development Capabilities
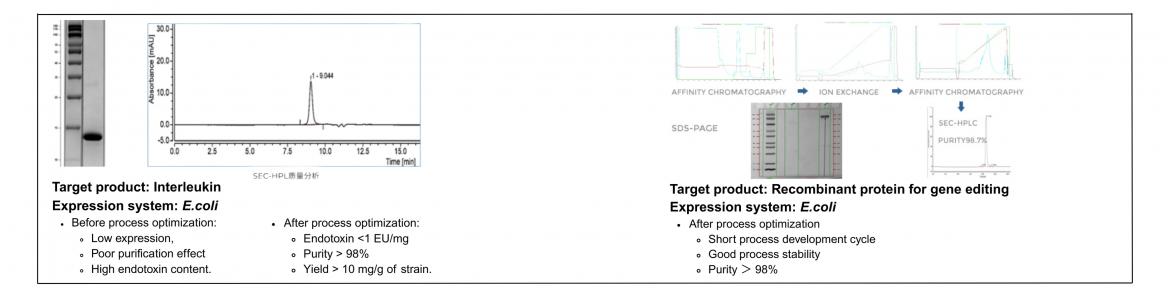
A successful approach to recombinant protein manufacturing hinges on an integrated process development strategy. Companies, including Yaohai Bio-Pharma, exemplify this by offering diverse experiences across various protein types, such as cytokines, VLPs, and allergens. Their tag-free purification platform significantly reduces process-related impurities, thereby enhancing product safety and efficacy. By defining Critical Quality Attributes (CQA) and Critical Process Parameters (CPP) through the Design of Experiments (DoE) methodology, businesses can effectively refine their processes to achieve desired outcomes while minimizing risks associated with high-concentration formulations.
Proven Success in Project Delivery
Management of protein process development can be daunting, especially with the complexity of scaling from R&D to commercial production. Yaohai Bio-Pharma’s proven record of success, hosting over 100 Chemistry, Manufacturing, and Controls (CMC) projects with a 99% project delivery rate, stands as a testament to their expertise. Their mature process development technology shortens development timelines, allowing clients to advance through clinical phases efficiently, from Phase I to Phase III, while complying with the highest global standards.
End-to-End Solutions and Efficient Collaboration
To support businesses in the recombinant protein manufacturing landscape, CRDMO solutions must embrace a holistic approach. With the ability to manage everything from early R&D to commercial production, Yaohai Bio-Pharma provides a dedicated team focused on integration and empowerment. Their commitment to cost-efficient innovation and seamless technology transfer plays a crucial role in helping companies scale their operations without compromising quality.
In summary, effective management of protein process development for high-concentration formulations requires robust collaboration and comprehensive capabilities. With a partner like Yaohai Bio-Pharma, businesses can navigate these complexities while achieving their goals in recombinant protein manufacturing.